Carbon Nanotube Injects Living Cells Without Damage
12 Jul, 2007 02:19 pm
Cell biologists have been trying to figure out the complicated processes happening inside a living cell. One commonly used approach is to introduce or inject probes, such as fluorescent molecules, into the cell?s interior. The difficulty has been to inject probes into the cells without damaging the cell membrane. Researchers have found a promising way by using carbon nanotubes--tiny tubes about 10,000 times thinner than a human hair made of carbon atoms.
As shown in Figure 1, the nanoinjector consists of a carbon nanotube attached to the tip of an atomic force microscope (AFM). AFM is conventionally used for imaging the surface topography of materials at the atomic scale. Integrated with an inverted fluorescence microscope, the AFM serves an ideal nanomanipulator for controlling the injection process with nanometer scale resolution. A linker molecule connects the designated cargo to the nanotube outer surface. Once the nanoneedle (CNT with cargo) penetrates the cell membrane, the linker was designed so that it was cleaved automatically by the environment inside the cell, liberating its attached cargo. The nanoneedle is then retracted by the AFM and the cargo is injected into the cell.
This nanoinjection was firstly demonstrated in cultured HeLa cells, which are human cervical epithelial cancer cells. The scientists attached protein-coated quantum dots onto CNT surface via the linker molecules. After the nanoneedle pokes into a targeted cell, the scientists observed clusters of quantum dots inside the cell using a fluorescence microscope. The scientists also used this technique to study the diffusion dynamics of the quantum dots inside the cell.
One of the advantages of this new technique is that the cell membrane is not damaged during the injection process, since the diameter of the carbon nanotube is comparable to the size of a protein. We used several assays to show that the nanoinjecton process does not reduce the cell viability, which you can not really achieve by using conventional techniques. says Chen who is a joint graduate student working with Professor Carolyn Bertozzi, a chemist and cell biologist, and Professor Alex Zettl, a physicist. Bertozzi adds, This is the first mechanism capable of delivering cargo into cells with nanometer-scale spatial control and no apparent cellular harm.
Another advantage is this nanoinjection technique does not use a carrier solvent, like some conventional methods. The carrier solvent can bring extra volume to the cell and thus damage the cells. states Chen, By eliminating the need of a carrier solvent, this technique can provide more flexibility for injecting probes into living cells. The researchers also argue that one of the beauties of the design is that a variety of linkers can be chosen to meet the specific requirements.
In the published study, the Berkeley team has already shown that they were able to deliver a discrete number of molecules to each cell's interior without the need of a carrier solvent and no discernible membrane or cell damage. In the future, they would like to combine the imaging capabilities of AFM and a confocal fluorescence microscope to carry out organelle-specific nanoinjections. With this capability, they could also introduce DNA and other biological molecules. In addition, parallel nanoinjections of different cell sites with different probes are currently under development.
Reference:
Xing Chen, et al. A cell nanoinjector based on carbon nanotubes. Proceedings of the National Academy of Sciences. 8218-8222, May 15, 2007, vol. 104.
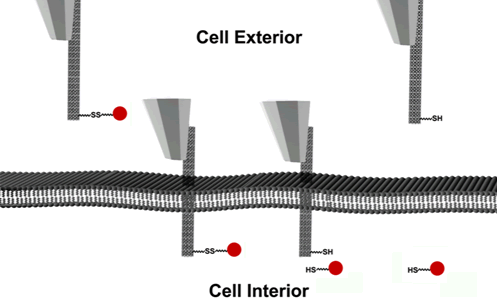
Figure 1. The schematic of the nanoinjector. An individual carbon nanotube is mounted on the tip of an atomic force microscope and the cargo is attached to the nanotube via a disulfide linker. After the needle penetrates the cell membrane, disulfide reduction within the cell releases the cargo. The injector is then retracted by AFM control, leaving cargo inside the cell.







A good report. The technology is in its infancy but the ability to enter cells without damage can allow precise manipulaton of cellular function.
IIt is an interesting report on utilizing carbon nanotubes in the absence of carrier solvent for releasing the required ?cargo? into the living cells without causing any harmful effect.